 Through trial and error, Thomson found he could get a “stronger” beam by shooting it through a positive anode with a hole in it. Thomson wondered if the air affected the results. Thomson knew that the cathode ray tubes that they had only work if there is a little air in the tube and the amount of air needed depended on the shape of the terminals. In 1896, Thomson wondered if there might have been something wrong with Hertz’s experiment with the two plates. ” This is why JJ Thomson was so confused, he felt that Perrin had, “conclusive evidence that the rays carried a charge of negative electricity” except that, “Hertz found that when they were exposed to an electric force they were not deflected at all.” What was going on? 
Perrin wrote, “the Faraday cylinder became negatively charged when the cathode rays entered it, and only when they entered it the cathode rays are thus charged with negative electricity. Then, in December of 1895, a French physicist named Jean Perrin used a magnet to direct a cathode ray into and out of an electroscope (called a Faraday cylinder) and measured its charge. Further, in 1892, Hertz accidentally discovered that cathode rays could tunnel through thin pieces of metal, which seemed like further proof that Crookes was so very wrong. Hertz decided that Crookes was wrong, if the cathode ray was made of charged particles then it should be attracted to a positive plate and repulsed from a negative plate.Įrgo, it couldn’t be particles, and Hertz decided it was probably some new kind of electromagnetic wave, like a new kind of ultraviolet light. However, 5 years later, a young German scientist named Heinrich Hertz found that he could not get the beam to move with parallel plates, or with an electric field. 
This gave a charismatic English chemist named William Crookes the crazy idea that the cathode ray was made of charged particles in 1879! Well, a cathode ray, or a ray in a vacuum tube that emanates from the negative electrode, can be easily moved with a magnet. Thomson discover the electron in 1897? Well, according to Thomson: “the discovery of the electron began with an attempt to explain the discrepancy between the behavior of cathode rays under magnetic and electric forces. 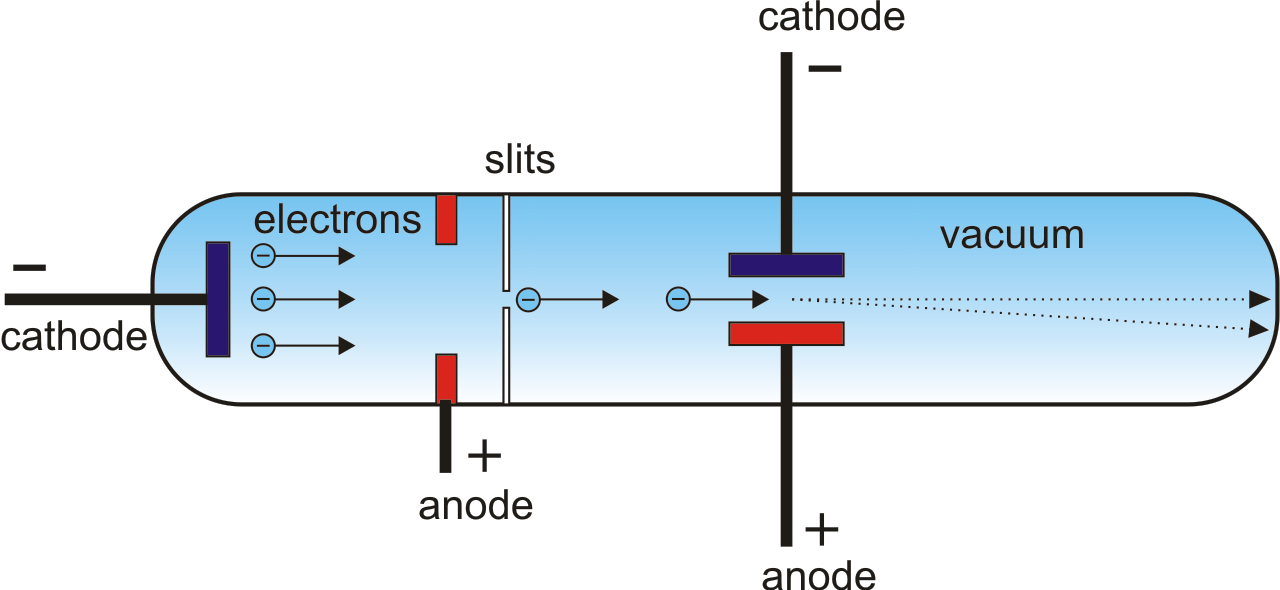
How Thomson Discovered Electrons: Trials and Errors In fact, a full eight of his research assistants and his son eventually earned Nobel Prizes, but, of course, like Thomson’s own Nobel Prize, that was in the future. He ended up having an unerring ability to pinpoint interesting phenomena for himself and for others. ” Suddenly, he had incredible resources, stability and ability to research whatever he wished. He mostly applied as a lark and was as surprised as anyone to actually get the position! “I felt like a fisherman who…had casually cast a line in an unlikely spot and hooked a fish much too heavy for him to land. In 1884, at the tender age of 28, Thomson applied to be the head of the Cavendish Research Institute. Instead, he relied on scholarships at universities – ironically leading him to much greater fame in academia. ” While in school, his father died, and his family didn’t have enough money for the apprenticeship. Thompson later recalled that, “the authorities at Owens College thought my admission was such a scandal – I expect they feared that students would soon be coming in perambulators – that they passed regulations raising the minimum age for admission, so that such a catastrophe should not happen again. 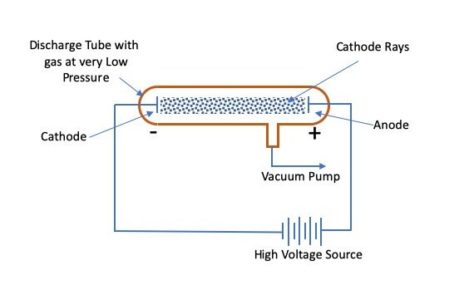
When he was 14 years old, Thomson planned to get an apprenticeship to a locomotive engineer but it had a long waiting list, so, he applied to and was accepted at that very young age to Owen’s college. Table of Contents The Start of JJ Thomson How Thomson Discovered Electrons: Trials and Errors Thomson’s Conclusion ReferencesĪ short history of Thomson: Joseph John Thomson, JJ on papers, to friends, and even to his own son, was born in Lancashire, England to a middle class bookseller. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
